Human LIF enzyme-linked immunoassay kit
| Specification | 96 Test |
|---|---|
| Sensitivity | 0.10 pg/ml (50 μl);0.36 pg/ml (10 μl) |
| Standard Curve Range | 0.69~500 pg/ml |
| Standard Curve Gradient | 7 Points/3 Folds |
| Number of Incubations | 2 |
| Detectable sample | Liquid phase sample of soluble substances. For example: serum, plasma, cell culture supernatant, tissue grinding liquid, etc. |
| Sample Volume | 50 μl/10 μl |
| Type | Fully Ready-to-Use |
| Operation Duration | 120min |
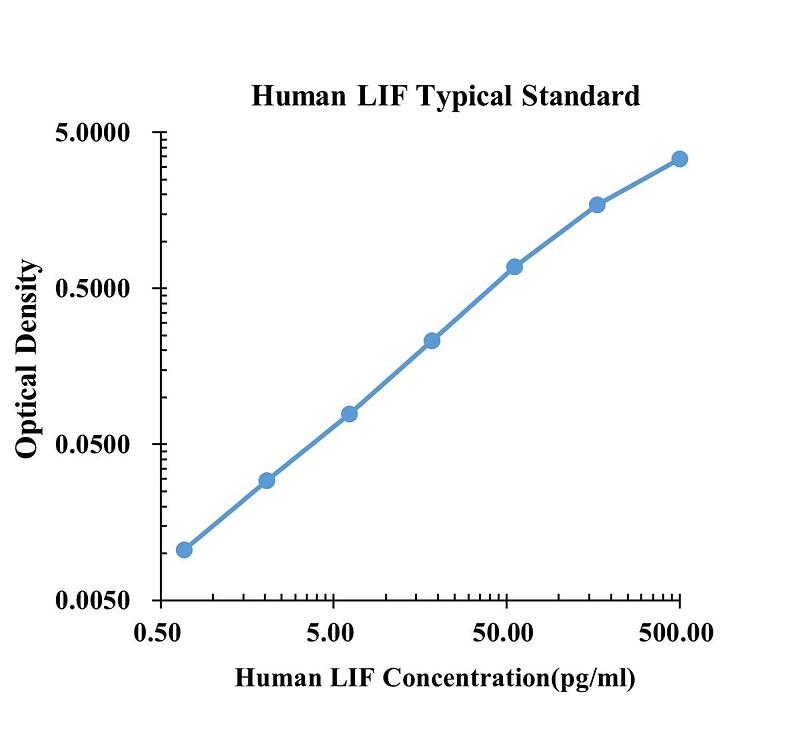
| pg/ml | O.D. | Average | Corrected | |
|---|---|---|---|---|
| 0.00 | 0.0142 | 0.0145 | 0.0143 | |
| 0.69 | 0.0244 | 0.0253 | 0.0248 | 0.0105 |
| 2.06 | 0.0432 | 0.0436 | 0.0434 | 0.0291 |
| 6.17 | 0.0940 | 0.0911 | 0.0926 | 0.0782 |
| 18.52 | 0.2485 | 0.2410 | 0.2448 | 0.2304 |
| 55.56 | 0.7105 | 0.6925 | 0.7015 | 0.6872 |
| 166.67 | 1.7840 | 1.6840 | 1.7340 | 1.7197 |
| 500.00 | 3.3730 | 3.4030 | 3.3880 | 3.3737 |
Precision
| Intra-assay Precision | Inter-assay Precision | |||||
| Sample Number | S1 | S2 | S3 | S1 | S2 | S3 |
| 22 | 22 | 22 | 6 | 6 | 6 | |
| Average(pg/ml) | 163.4 | 48.5 | 10.5 | 177.7 | 61.2 | 12.2 |
| Standard Deviation | 9.6 | 2.3 | 0.6 | 5.4 | 2.3 | 0.6 |
| Coefficient of Variation(%) | 5.9 | 4.8 | 6.2 | 3.0 | 3.8 | 4.9 |
Intra-assay Precision (Precision within an assay) Three samples of known concentration were tested twenty times on one plate to assess intra-assay precision.
Inter-assay Precision (Precision between assays) Three samples of known concentration were tested six times on one plate to assess intra-assay precision.
Spike Recovery
The spike recovery was evaluated by spiking 3 levels of human LIF into health human serum sample. The un-spiked serum was used as blank in this experiment.
The recovery ranged from 93% to 116% with an overall mean recovery of 102%.
Sample Values
| Sample Matrix | Sample Evaluated | Range (pg/ml) | Detectable (%) | Mean of Detectable (pg/ml) |
|---|---|---|---|---|
| Serum | 30 | 1.85-16.87 | 100 | 4.94 |
Serum/Plasma – Thirty samples from apparently healthy volunteers were evaluated for the presence of VE Cadherin in this assay. No medical histories were available for the donors.
Product Data Sheet
Background: LIF
LIF (leukemia inhibitory factor) is a widely expressed, highly and variably glycosylated, 32‑62 kDa, monomeric, pleiotropic cytokine in the IL‑6 family of helical cytokines. The first exon encoding the signal sequence is alternately spliced, resulting in LIF-D, LIF-M, and LIF‑T mRNAs that produce secreted, extracellular matrix‑associated, and intracellular forms, respectively. LIF-D and LIF-M mRNAs produce identical 180 amino acid (aa) mature sequences. Mature human LIF (180 aa) shares 78%, 82%, 91%, 88 and 87% aa sequence identity with mouse, rat, canine, bovine, and porcine LIF, respectively. The LIF receptor is a heterodimer of a type I transmembrane ligand‑binding subunit, LIFR (gp190), and the type I transmembrane signal transducing subunit, gp130, signaling especially through STAT3 and JAK kinases. Gp130 and members of the LIFR family also mediate the biological effects of Oncostatin M, Cardiotrophin‑1, Galectin‑10, CNTF, IL‑6,
IL‑11, and IL‑27. A soluble LIFR has been reported in the mouse. Depending on the cells and their context, LIF either opposes or favors differentiation. LIF produced by the uterine endometrium supports successful implantation of the embryo, promotes proliferation and maintenance of pluripotency in embryonic stem cells, and favors proliferation of progenitor cell types such as hematopoietic stem cells. However, excess LIF blocks differentiation of embryoid bodies, indicating the importance of LIF regulation. LIF is produced by CD4+ T cells in response to activation, and is required by the thymic epithelium to support T cell maturation. LIF expression is up‑regulated by neuronal injury, and promotes motor neuron survival and oligodendrocyte myelination. LIF is produced by the adrenal cortex and likely enhances its production of cortisol and aldosterone. LIF can function as an autocrine growth factor in some pancreatic cancers, but induces differentiation in the myeloid leukemic cell line M1. Tumor LIF can also induce formation of immunosuppressive tumor‑associated macrophages. LIF promotes endometrial remodeling and differentiation of adipocytes and cardiac smooth muscle cells. It promotes regulatory T cell and inhibits Th17 cell differentiation, thus promoting tolerance, down‑regulating inflammation, and contributing to immune tolerance during pregnancy and in the nervous system.

